Passenger Cars, SUVs, and Light-Duty Trucks
Catalytic Converters – Three-Way Catalytic Converter
The three-way converter (TWC) has been the primary emission control technology on light-duty gasoline vehicles since the early 1980s. The use of TWCs, in conjunction with an oxygen sensor-based, closed-loop fuel delivery system, allows for simultaneous conversion of the three criteria pollutants, HC, CO, and NOx, produced during the combustion of fuel in a spark-ignited engine. The active catalytic materials are present as a thin coating of precious metal (e.g., Pt, Pd, Rh), and oxide-based inorganic promoters and support materials on the internal walls of the honeycomb substrate. The substrate typically provides a large number of parallel flow channels to allow for sufficient contacting area between the exhaust gas and the active catalytic materials without creating excess pressure losses. Although the primary components and function of a TWC has remained relatively constant during its more than twenty years of use on light-duty gasoline vehicles, each of the primary converter components (catalytic coating, substrate, mounting materials) has gone through a continuous evolution and redesign process in order to improve the overall performance of the converter while maintaining a competitive cost effectiveness of the complete assembly.

Figure. TWC
Catalytic Converters – Diesel Oxidation Catalyst
In most applications, a diesel oxidation catalyst consists of a stainless steel canister that contains a honeycomb structure called a substrate or catalyst support. There are no moving parts, just large amounts of interior surface area. The interior surfaces are coated with catalytic metals such as platinum or palladium. It is called an oxidation catalyst because the device converts exhaust gas pollutants into harmless gases by means of chemical oxidation. In the case of diesel exhaust, the catalyst oxidizes CO, HC, and the liquid hydrocarbons adsorbed on carbon particles. In the field of mobile source emission control, liquid hydrocarbons adsorbed on the carbon particles in engine exhaust are referred to as the soluble organic fraction (SOF) — the soluble part of the particulate matter in the exhaust. Diesel oxidation catalysts are efficient at converting the soluble organic fraction of diesel particulate matter into carbon dioxide and water. DOCs can typically achieve 25 to 40% particulate reduction by simply burning the SOF component of particulate matter.
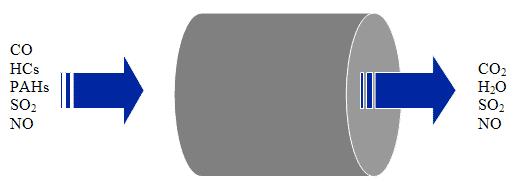
Figure. DOC
Catalytic Converters – SCR System
A Selective Catalytic Reduction (SCR) system uses a metallic or ceramic wash-coated catalyzed substrate, or a homogeneously extruded catalyst and a chemical reductant to convert nitrogen oxides to molecular nitrogen and oxygen in oxygen-rich exhaust streams like those encountered with diesel engines. In mobile source applications, an aqueous urea solution is usually the preferred reductant. Upon thermal decomposition in the exhaust, urea decomposes to ammonia which serves as the reductant. In some cases ammonia has been used as the reductant in mobile source retrofit applications. As exhaust and reductant pass over the SCR catalyst, chemical reactions occur that reduce NOx emissions to nitrogen and water. SCR catalysts can be combined with a particulate filter for combined reductions of both PM and NOx.
Open loop SCR systems can reduce NOx emissions by 75 to 90 percent. Closed loop systems on stationary engines can achieve NOx reductions of greater than 95 percent. SCR systems are also effective in reducing HC emissions up to 80 percent and PM emissions 20 to 30 percent. Like all catalyst-based emission control technologies, SCR performance is enhanced by the use of low sulfur fuel.
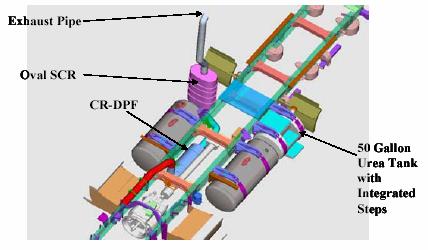
Figure. SCR System
Catalytic Converters – Lean NOx Catalyst
Controlling NOx emissions from a diesel engine is inherently difficult because diesel engines are designed to run lean. In the oxygen-rich environment of diesel exhaust, it is difficult to chemically reduce NOx to molecular nitrogen. The conversion of NOx to molecular nitrogen in the exhaust stream requires a reductant (HC, CO or H2) and under typical engine operating conditions, sufficient quantities of reductant are not present to facilitate the conversion of NOx to nitrogen.
Some lean NOx catalyst (LNC) systems inject a small amount of diesel fuel or other reductant into the exhaust upstream of the catalyst. The fuel or other hydrocarbon reductant serves as a reducing agent for the catalytic conversion of NOx to N2. Other systems operate passively without any added reductant at reduced NOx conversion rates. A lean NOx catalyst often includes a porous material made of zeolite (a micro-porous material with a highly ordered channel structure), along with either a precious metal or base metal catalyst. The zeolites provide microscopic sites that are fuel/hydrocarbon rich where reduction reactions can take place. Without the added fuel and catalyst, reduction reactions that convert NOx to N2 would not take place because of excess oxygen present in the exhaust. Currently, peak NOx conversion efficiencies typically are around 10 to 30 percent (at reasonable levels of diesel fuel reductant consumption).

Figure. LNC
Catalytic Converters – Lean NOx Trap
Another type of catalyst being developed for diesel engines are known as lean NOx traps (LNT) because they function by trapping the NOx in the form of a metal nitrate during lean operation of the engine. The most common compound used to capture NOx is Barium Hydroxide or Barium Carbonate. Under lean air to fuel operation, NOx reacts to form NO2 over a platinum catalyst followed by reaction with the Barium compound to form BaNO3. Following a certain amount of lean operation, the trapping function will become saturated and must be regenerated. This is commonly done by operating the engine in a fuel rich mode for a brief period of time to facilitate the conversion of the barium compound back to a hydrated or carbonated form and giving up NOx in the form of N2 or NH3. LNT catalyst can be combined with a zeolite based SCR catalyst to trap ammonia and further reduce NOx via a selective catalytic reduction reaction to nitrogen.
Particulate Filters – Diesel Particulate Filter
Diesel particulate filters remove particulate matter found in diesel exhaust by filtering exhaust from the engine. In order to meet the stringent particulate emissions that are required for diesel light duty vehicles starting with the 2007 model year, the highest efficiency particulate filter is required. These are commonly made from ceramic materials such as cordierite, aluminum titanate, mullite or silicon carbide. The basis for the design of wall flow filters is a honeycomb structure with alternate channels plugged at opposite ends. As the gasses passes into the open end of a channel, the plug at the opposite end forces the gasses through the porous wall of the honeycomb channel and out through the neighboring channel. The ultrafine porous structure of the channel walls results in greater than 90% percent collection efficiencies of these filters. Wall flow filters capture particulate matter by interception and impaction of the solid particles across the porous wall. The exhaust gas is allowed to pass through in order to maintain low pressure drop.
Since a filter can fill up over time by developing a layer of retained particles on the inside surface of the porous wall, engineers that design engines and filter systems must provide a means of burning off or removing accumulated particulate matter and thus regenerating the filter. A convenient means of disposing of accumulated particulate matter is to burn or oxidize it on the filter when exhaust temperatures are adequate. By burning off trapped material, the filter is cleaned or “regenerated” to its original state. The frequency of regeneration is determined by the amount of soot build-up resulting in an increase in back pressure. To facilitate decomposition of the soot, a catalyst is used either in the form of a coating on the filter or a catalyst added to the fuel. Filters that regenerate in this so-called “passive” fashion cannot be used in all situations. The experience with catalyzed filters indicates that there is a virtually complete reduction in odor and in the soluble organic fraction of the particulate. Despite the high efficiency of the catalyst, a layer of ash may build up on the filter requiring replacement or servicing. The ash is made up of inorganic oxides from the fuel or lubricants used in the engine and will not decompose during the regular soot regeneration process. In some applications or operating cycles, the exhaust never achieves a high enough temperature to completely oxidize the soot even in the presence of a catalyst. In these instances, an “active” regeneration system must be employed. Active regeneration utilizes a fuel burner or a resistively heated electric element to heat the filter and oxidize the soot. Active regeneration can be employed either in-place on the vehicle or externally. During external regeneration, the filter is removed from the vehicle and heated in a controlled chamber.
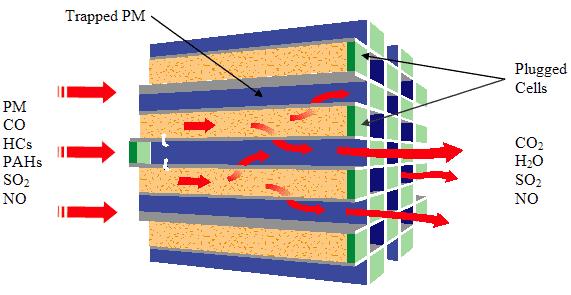
Figure. DPF
Sensor Technologies – Temperature Sensor
Temperature sensors are used for two purposes: The first is as a warning system, typically on obsolete oxidation-only catalytic converters. The function of the sensor is to warn of temperature excursions above the safe operating temperature of the catalytic converter. However, modern catalytic converters are not as susceptible to temperature damage. Many modern three-way Platinum-based converters are able to handle temperatures of 900 degrees C sustained, while many modern three-way Palladium-based converters are able to handle temperatures of 925 degrees C sustained. Temperature sensors are also used to monitor the temperature rise over the catalytic converter core.
Sensor Technologies – Oxygen Sensor
Oxygen sensors are part of the closed loop fuel feedback control system, associated with modern three-way catalyst emission control systems on gasoline engines. The closed loop fuel feedback control system is responsible for controlling the air/fuel ratio of the catalytic converter feed gas. During the closed loop operation, the electronic control module (ECM) keeps the air/fuel ratio adjusted to around the ideal 14.7 to 1 ratio. Signal from the oxygen sensor is used to determine the exact concentration of oxygen in the exhaust stream. From this signal, the ECM determines whether the mixture is richer or leaner than the ideal 14.7 to 1 air/fuel ratio. If the air/fuel ratio deviates from its preprogrammed swings, catalyst efficiency decreases dramatically, especially for NOx reduction. The oxygen sensor informs the ECM of needed adjustments to injector duration based on exhaust conditions. After adjustments are made, the oxygen sensor monitors the correction accuracy and informs the ECM of additional adjustments. The oxygen sensor is also an integral part of the onboard diagnostic (OBD) system which monitors the proper functioning of the emission control system of the vehicle. If the sensor detects oxygen content of the exhaust that is outside the specified range of the engine calibration, it will trigger the engine light to come on in the instrument cluster.
Sensor Technologies – NOx Sensor
NOx sensors represent state of the art technology that can be applied to gasoline lean burn engines as part of a broader engine control or diagnostic system used to insure proper operation of the NOx emission control system. These sensors can be incorporated independent of the NOx emission control technology used on the vehicle and their function is primarily to monitor the NOx conversion efficiency of the catalyst. The sensors can work as part of a feedback loop to the control unit on the emissions system to make real time adjustments and optimize NOx conversion. The principle of operation of one type of NOx sensor is based on proven solid electrolyte technology developed for oxygen sensors. The dual chamber zirconia sensing element and electro-chemical pumps work in conjunction with precious metal catalyst electrodes to control the oxygen concentration within the sensor and convert the NOx to nitrogen. The sensor sends output signals in volts that are directly proportional to ppm NOx concentration. The sensors can be incorporated upstream and downstream of the catalyst, for example, to provide a feedback control loop to the ECU of the emissions system. The ECU can than make adjustments to optimize NOx conversion performance. In the case of SCR technology, feedback can also be provided to the urea dosing system whereas in the case of lean NOx trap catalysts, a feedback loop could signal the regeneration of the trap.
Thermal Management Strategies
The majority of emissions from today’s gasoline and diesel engines occur during cold start before the catalyst can achieve optimum operating temperatures. Exhaust system manufacturers have been working together with catalyst companies to develop ways to heat up the catalyst as quickly as possible. The greatest impact came from the introduction of close coupled catalysts (CCC) to supplement the existing underfloor systems in the mid-1990. This positioned a smaller catalytic converter close to the exhaust manifold to allow rapid oxidation of CO and hydrocarbons. The exothermic heat generated in the CCC by these reactions facilitates the rapid heat up of the down stream, larger, underfloor, TWC. In later developments, the CCC was sometimes formulated to be a fully functional TWC with the underfloor unit serving as a clean-up catalyst to convert the final 10-20% of the pollutants.
The beneficial impact on reducing cold start emissions via thermal management has led to numerous improvements to the exhaust system components up stream of the converter in order to retain as much heat as possible in the exhaust gases. Manufacturers have developed ways to insulate the exhaust manifold and exhaust pipe. Attaching the CCC to a double walled, stainless steel exhaust pipe containing an air gap within the tube walls is probably the most common thermal management strategy used today. To meet the tightest SULEV and PZEV regulations required attention to the temperature distribution at the face of the CCC. This led to new inlet cone designs and modification to the shape of the space in front of the close coupled substrate.
Engine/Fuel Management
Achieving near-zero exhaust emission targets requires a systems approach. Engine manufacturers are focusing on ways to control engine operation to reduce engine out emissions as low as possible and reduce the burden on the catalysts.
Approaches aimed at reducing cold start emissions involve retarding the ignition timing so as to allow some hydrocarbons to pass through in the exhaust and light off the catalyst sooner. Variable valve timing (VVT) is being used to introduce some fraction of exhaust gas into the combustion process and reduce HC and NOx emissions. On clean diesel engines, Exhaust Gas Recirculation (EGR) is used to dilute intake air with some fraction of exhaust gas to lower the combustion temperatures resulting in lower engine out NOx emissions.
Direct injection of fuel into the cylinders rather than port injection has allowed better control of the air fuel ratio during combustion and resulted in better fuel utilization. Improved turbulence and mixing in the intake port of some low emission engines have resulted in a 24% fuel savings. Clean diesel engines have benefited significantly from common rail fuel injection which allows for electronically controlled injection at very high pressures. Through the use of pilot and retarded injection strategies or in combination with injection rate shaping clean diesels have achieved significant reduction in NOx over conventional diesel injection such as pipe-line or unit injection. Common rail and electronic injection control is very effective in carefully controlling post injection of fuel making it suitable for use with emission control devices such as particulate filters, NOx adsorbers and lean NOx catalysts requiring brief periods of fuel rich exhaust to facilitate regeneration of the catalyst or filter.
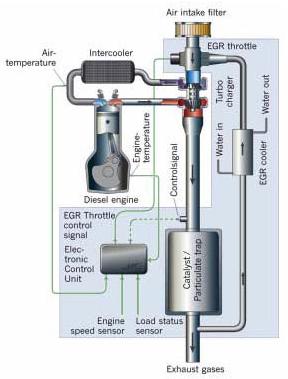
Figure. Low-pressure EGR + DPF
Evaporative Emission Controls
The purpose of evaporative emissions systems is to reduce or eliminate the release of vaporized HC into the atmosphere. The HC vapors, such as volatile organic compounds (VOCs), react in the atmosphere with nitrogen oxides (NOx) and contribute to the formation of ground level ozone and photochemical smog. Ground-level ozone is an air pollutant with harmful effects to plants, human respiratory systems, and an irritant to our eyes.
The major source of evaporative emissions is from the fuel system and, therefore, it is not surprising that fuel injection technology provides significant evaporative emissions benefits. The second major source of evaporative emissions comes from the positive crankcase ventilation (PCV) system. PCV systems collect and consume fuel that is pushed by the piston rings during the compression and power strokes in a 4-stroke (4-cycle) engine. These systems have been used on automobiles since the 1960s in the form of PCV valves. Evaporative emissions regulations have been in effect for automobiles since 1970 in California. Fuel injection systems effectively eliminated the vaporization of fuel from open carburetors. Evaporative emission control systems on cars have increased in complexity over the years to achieve the extremely low evaporative emissions (0.054g/day) from the fuel system as required by California’s Partial Zero Emission Vehicle (PZEV) “zero” evaporative emission limits. Other significant sources of evaporative emissions from the fuel system include permeation of the fuel tank and fuel delivery hoses.
Types of evaporative emissions are classified into five categories:
- Diurnal: This represents gasoline that evaporates due to the rise in ambient temperature.
- Running losses: Represent gasoline that vaporizes due to the heat of the engine and exhaust system during normal operation.
- Resting losses: Natural permeation that occurs from the fuel delivery system while not operating under ambient conditions.
- Hot Soak: Vaporization of fuel due to the retained heat of the engine after the engine is turned off.
- Refueling: Represents the fuel vapors that escape from the tank by the displacement of liquid fuel.
Evaporative emissions are measured using a sealed housing for evaporative determination (SHED) apparatus over the course of a multi-day Federal Test Procedure (FTP) to quantify all of the various forms of evaporative emissions. This testing is generally most effective in determining diurnal and hot soak emissions.
In addition to low permeation hoses, fuel tanks, and seals, evaporative emission controls consists of carbon canisters connected to the fuel system to capture and recycle HC vapors back to the intake of the engine to be consumed as fuel. The carbon is a high-surface area, pelletized material that adsorbs fuel vapors via loose chemical bonds and releases them in a controlled fashion via a purge solenoid. The purge solenoid is activated by the on-board control module when the engine’s control system is operating in “closed” loop fuel control. Carbon materials or other materials that adsorb vapor phase hydrocarbons such as zeolites can also be displayed in the engine’s air intake system to control evaporative emissions associated with fuel leakage from fuel injectors when the engine is not running (see Figure below).
Metal fuel tanks exhibit no permeation; however, the need for lighter weight and complex shapes has led to the development of low permeation plastic tanks. In this case, gas tanks are made of layered polymers and blends that reduce tank permeation by 95% compared to traditional plastic fuel tanks. Similar types of thermo-polymers are molded as thin (0.1 mm) layers in the inside of fuel lines to achieve similar fuel permeation reductions. A vented fuel cap serves to allow air to enter as fuel is depleted while venting expanded vapors in the fuel tank into the carbon canister.
The combination of carbon canisters, the use of low permeable polymers, and fuel injection systems has been demonstrated as a very effective evaporative emissions control strategy. Regulatory agencies around the world are continuing to apply evaporative emission limits to a broad range of on-road and off-road vehicles and engines including motorcycles, small garden equipment, and marine applications.
In the figure below, carbon canisters come in many shapes and sizes, from advanced multi-stage PZEV units (A), to canisters designed for marine (B), to motorcycle and small engine applications (C). (D) is an integrated device to be added to a LEV II evap system/canister vent port to make it a PZEV evap system/canister.
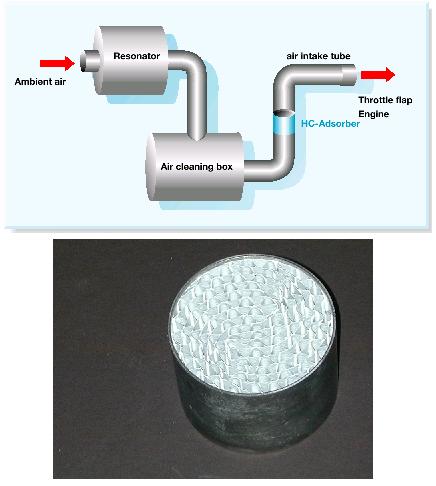
Figure. Air intake hydrocarbon adsorber coated on metal monolith and incorporated into air intake system
Enhanced Combustion Technologies
Understanding and controlling the combustion process is the first step in reducing engine out emissions and reducing the burden on the emission control systems within the exhaust. Engine design is an important part of controlling and facilitating the combustion process.
The most dramatic approach that has been used in recent years on spark-ignited engines is the development of gasoline direct injection (GDI) engines. Early fuel injected engines used a port injection approach where fuel was injected into a port to allow it to evaporate and mix uniformly with the air. This provides little control over the air and fuel mixture entering the cylinder. GDI engines inject the fuel directly into the combustion chamber allowing varying injection strategies depending on engine load. At high load, fuel is injected into the engine early during the induction stroke giving a stoichiometric or rich air/fuel ratio. A GDI engine can also operate in an ultra lean combustion mode during cruising situations when little acceleration is required. In this case the fuel is injected in the latter stages of the compression stroke just prior to ignition. This allows a small amount of fuel to be placed near the spark plug. The effective air fuel ratio is very lean resulting in significant fuel savings. The reason that partial lean burn GDI engines have not reached broad application in the market is the difficulty in meeting NOx emissions regulation during long periods of lean operation. GDI engines can also be designed for stoichiometric operation and make use of three-way catalysts for lowering exhaust emissions.
In diesel engines, controlling combustion is the key approach to reducing engine out particulate emissions by optimizing the mixing between the fuel and air. Some common ways to increase mixing is through combustion chamber modifications to facilitate turbulent flow as well as fuel injector design to modify the spray pattern. Variable geometry turbocharging (VGT), which delivers variable quantities of pressurized air based on driving conditions, has been effective in reducing PM emissions by maintaining lean combustion in the engine. Reducing compression ratios have been shown effective in reducing combustion temperatures and in turn NOx emissions. Some common approaches to enhance air turbulence and improve fuel distribution within the cylinders include improvements to the design of fuel injectors, combustion chambers and injection ports. Some engine manufacturers have been able to achieve improvements to the combustion during cold start by making modifications to the design of intake air control valves resulting in a 40-50% reduction in HC emissions and injection ports among others.
